

Cell 133, April 18, 2008
GREGG L.SEMENZA, MARY K.NEJFELT, SUZIE M.CHI, AND STYLIANOS E.ANTONARAKIS
Gregg L. Semenza1,2,*
1Vascular Program, Institute for Cell Engineering; Departments of Pediatrics, Medicine, Oncology, and Radiation Oncology
2McKusick-Nathans Institute of Genetic Medicine, The Johns Hopkins University School of Medicine
Baltimore, MD 21205, USA
*Correspondence: gsemenza@jhmi.edu
DOI 10.1016/j.cell.2008.04.004
The transcription factor HIF-1 mediates adaptive responses to hypoxia, and its activity is negatively regulated by O2-dependent binding of the von Hippel-Lindau (VHL) protein. In this issue, Boutin et al. (2008) use conditional knockout mice to demonstrate that sensing of O2 by keratinocytes in the epidermis leads to alterations in cutaneous blood flow that affect the production of the hormone erythropoietin, thereby modulating red blood cell production and the O2-carrying capacity of blood.
The physiology of modern day mammals has been shaped by natural selection over the past several billion years, during which life on Earth has thrived in an O2-rich environment. Metazoan species evolved a key protein, the transcription factor hypoxia-inducible factor 1 (HIF-1), to regulate O2 delivery to tissues. Decreased O2 in the blood of mammals is sensed by the carotid body, which is located at the bifurcation of the common carotid artery and activates the cardiovascular and respiratory centers in the central nervous system to increase ventilation and cardiac output (Kline et al., 2002). A reduction in O2 is also sensed by cells in the liver and kidney, which respond by producing erythropoietin (EPO), the hormone that controls red blood cell production and, thus, the O2-carrying capacity of blood (Stockmann and Fandrey, 2006). HIF-1, originally discovered as the factor that mediates hypoxia-induced transcription of the human EPO gene (Semenza and Wang, 1992), is also required for O2 sensing by the carotid body (Kline et al., 2002). HIF-1 is composed of two subunits: constitutively expressed HIF-1β and O2-regulated HIF-1α (Wang et al., 1995). Database searches led to the identification of HIF-2α, which is also regulated by O2, dimerizes with HIF-1β, and regulates an overlapping but distinct battery of target genes (Elvidge et al., 2006; Patel and Simon, 2008). Under normoxic conditions, HIF-1α and HIF-2α are subjected to hydroxylation on proline residues (402 and 564 in human HIF-1α). The modification is required for the binding of the von Hippel-Lindau (VHL) tumor suppressor protein, the recognition component of an E3 ubiquitinprotein ligase that targets HIF-1α for proteasomal degradation (Kaelin, 2005; Schofield and Ratcliffe, 2005). Under hypoxic conditions, hydroxylation is inhibited and the VHL protein does not bind to HIF-1, leading to the accumulation of HIF-1α and HIF-2α.
Homozygous deletion in the mouse germline of the Hif-1α or Vhl locus results in embryonic lethality. Using Crelox technology to generate conditional knockout mice at these loci in specific cell types and at specific time points in postnatal life has provided insights into complex homeostatic mechanisms, which may not have been possible using other approaches. That has certainly proven to be the case with the deletion of these genes in mouse epidermal cells (keratinocytes) as Randy Johnson and colleagues report in this issue (Boutin et al., 2008). They show that deletion of the Vhl gene in mouse keratinocytes results in increased EPO levels in plasma and development of polycythemia, a condition in which too many red blood cells are produced and blood viscosity increases. Interestingly, humans with an inherited form of polycythemia are homozygous for a germline missense mutation that reduces, but does not eliminate, VHL activity (Ang et al., 2002). But given that EPO is not expressed in the skin, how does loss of the VHL protein in mouse keratinocytes result in increased production of EPO and red blood cells?
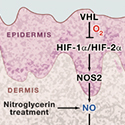
The skin is perfused by capillaries that are located in the dermis (Figure 1). Regulation of cutaneous blood flow is an important component of the homeostatic mechanism that maintains the human core body temperature at 37°C despite wide fluctuations in environmental temperature. Vasodilation of dermal capillaries increases heat loss, whereas vasoconstriction prevents it. As Boutin et al. (2008) now show, deletion of VHL in mouse keratinocytes causes a marked increase in cutaneous blood flow and dysregulated expression of HIF-1α and especially HIF-2α. This results in the expression of HIF-1-regulated genes, including Nos2, which encodes inducible nitric oxide synthase. The expression of Nos2 leads to increased generation of nitric oxide (NO), a potent vasodilator. Cutaneous vasodilation, that is, more blood flow to the skin, results in less blood flow to other organs. In the liver, blood flow is sufficiently reduced to result in tissue hypoxia, a condition that induces expression of the Epo gene. Consistent with this mechanism for increased EPO production, Boutin et al. did not observe elevated EPO expression in the keratinocyte-specific Vhl knockout mice that also lacked either HIF-2α (in keratinocytes) or NOS2 (in all cells). Remarkably, transdermal administration of nitroglycerin (an NO donor) to wild-type mice boosted plasma levels of EPO, whereas systemic treatment did not. Thus, the homeostatic mechanism for maintaining the O2-carrying capacity of blood can be disrupted by the dysregulation of cutaneous blood flow (Figure 1). But do these intriguing observations have any relevance for normal mammalian physiology?
To answer this question, the authors performed two experiments, one mundane and the other insane. In the mundane experiment, they eliminated the expression of either HIF-1α or HIF-2α in mouse keratinocytes expressing wild-type VHL and analyzed the effect on hypoxia-induced EPO production. One might predict that keratinocyte-specific loss of HIF-2α would impair EPO production in response to hypoxia, but surprisingly, loss of HIF-2α had no effect. But when mice with a keratinocyte-specific deletion of HIF-1α were exposed to hypoxia, the predicted increase in plasma EPO levels was blunted and induction of EPO expression in the kidney was lost. These results suggest that physiological regulation of cutaneous blood flow in response to changes in atmospheric O2 is mediated by HIF-1α, whereas the pathological dysregulation resulting from VHL loss-of- function is primarily mediated by HIF-2α (Figure 1). Taken together, these results support the conclusion that HIF-1 activity in the skin is important for EPO production by the kidney in response to hypoxia.